搜索


 Time:Jun 12, 2025
Time:Jun 12, 2025
Alzheimer's disease (AD), as the foremost neurodegenerative disorder, presents with memory decline and cognitive impairment as its core clinical symptoms. Its pathological hallmarks include cerebral grey matter atrophy, amyloid-β (Aβ) aggregation forming plaques, and tau protein forming neurofibrillary tangles. Positron emission tomography (PET) brain imaging targeting Aβ and tau pathological aggregates serves as the gold standard for AD clinical diagnosis. Despite sharing common pathological features, clinical presentations among AD patients exhibit significant variability. Recent research indicates this heterogeneity may be closely linked to structural diversity within Aβ fibrillar aggregates: Type I fibrillar aggregate subtypes predominate in sporadic AD brains, whereas Type II subtypes are more prevalent in familial AD. Furthermore, novel Aβ fibrillar conformational subtypes distinct from Types I and II have been identified in AD brains with different genetic backgrounds (e.g., Arctic mutation and Down syndrome). The complex pathological heterogeneity exhibited by AD patients poses challenges to the practical scope and accuracy of existing molecular diagnostic methods, such as PET brain imaging targeting Aβ aggregates. Furthermore, the specific molecular mechanism by which Aβ aggregate PET tracers, long used clinically, target and recognise Aβ aggregates, along with their selectivity patterns for different Aβ aggregate subtypes, remain unknown. This severely hinders the development of next-generation Aβ PET probes.
On 11 June 2025, the research team led by Professor Li Dan at the Zhangjiang Advanced Research Institute of Shanghai Jiao Tong University, in collaboration with Professor Liu Cong's team at the Centre for Interdisciplinary Research in Biology and Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, and Professor Qiu Wenying's team at the Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences, published a study titled ‘Unravelling Alzheimer's Complexity with a Distinct Aβ42 Fibril Type and Specific AV-45 Binding’ in Nature Chemical Biology. This work identified a novel pathological fibrillar aggregate type (Type III) of Aβ42 within the ‘soluble fraction’ of AD patient brain tissue. It also elucidated for the first time the molecular mechanism by which the clinical PET tracer AV-45 recognises Aβ42. This provides both theoretical foundations and novel approaches for the precise molecular subtyping of AD and the development of next-generation PET tracers.
Pathological amyloid fibrillar aggregates can typically be isolated from AD patient brain tissue samples via biochemical methods. Due to their highly stable hydrophobic core, they are generally believed to reside within the insoluble fraction extracted using the detergent Sarkosyl, whereas the soluble fraction is thought to predominantly contain oligomers or protofibrils. In this study, researchers identified substantial quantities of Aβ42 and TMEM106B fibrillar aggregates within the soluble fraction isolated from AD patient brain tissue. These aggregates exhibited significantly looser structural organisation compared to fibrils in the insoluble fraction. Notably, even when fibrillar aggregates within both fractions share identical atomic structures (e.g., Type I fibrils across different fractions), differing surface chemical modifications may directly cause variations in their sedimentation properties. This explains why the same fibril type can coexist within both soluble and insoluble fractions. Moreover, the research team identified for the first time a novel Aβ42 fibrillar aggregate type—Type III—exclusive to the soluble fraction. This fibril exhibits a hybrid conformation of Type I and Type II. This discovery offers new insights into understanding the compositional and structural complexity of pathological aggregates in AD, as well as enabling precise AD subtyping based on different Aβ42 subtype structures.
Researchers further investigated the interaction mechanisms between different Aβ fibril aggregate subtypes and the clinically widely used Aβ aggregate PET tracer AV-45 (Florbetapir, trade name Amyvid). Cryo-EM structural analysis revealed its complex with Aβ42 Type I and Type III fibrils. The study revealed that within Type I fibrils, AV-45 vertically embeds into a helical core channel formed by β-sheet layers. This unique binding pattern differs from the conventional binding modes of small-molecule ligands observed with pathological amyloid fibrils. Type III fibrils, lacking a core channel analogous to Type I, exhibited no electron density indicative of AV-45 binding within the fibril core.
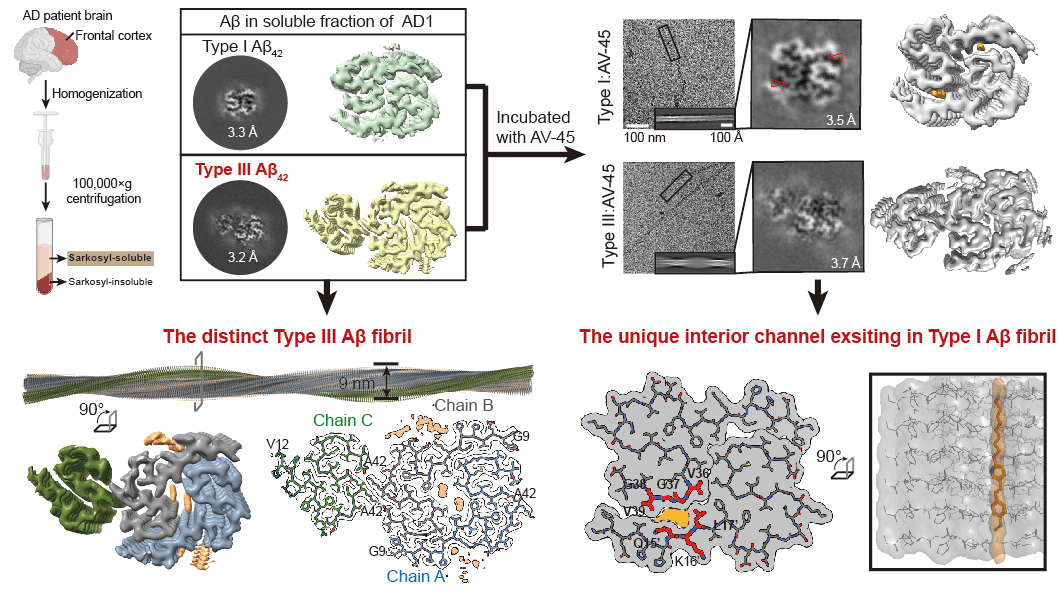
Figure 1. Discovery of novel Aβ42 fibrillar aggregate types and the binding mechanism between aggregates and AV-45.
In summary, methodologically, this study redefines the pathological significance of the ‘soluble fraction’ in Sarkosyl extraction, offering new avenues for comprehensively understanding the structural diversity of pathological amyloid fibrillar aggregates. pathologically, it reveals the potential link between Aβ42 fibril structural heterogeneity and clinical disease manifestations, laying foundations for precise AD subtype classification; clinically, it elucidates AV-45's specific binding mechanism, providing a structural blueprint for developing novel tracers and targeted therapies against distinct fibril types.
Professor Dan Li from the Zhangjiang Advanced Research Institute at Shanghai Jiao Tong University, Researcher Cong Liu from the Centre for Interdisciplinary Research in Biology and Chemistry at the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, and Professor Wenying Qiu from the Institute of Basic Medical Sciences, Chinese Academy of Medical Sciences & Peking Union Medical College served as corresponding authors. PhD candidate Qinyue Zhao and Dr Youqi Tao, a graduate of the Bio-X Institute at Shanghai Jiao Tong University, are the co-first authors of this paper. Professor Ma Chao from Peking Union Medical College, Professor Shen Yong and Professor Gao Feng from the University of Science and Technology of China, along with patients and their families, provided invaluable brain tissue samples for this research. Professor Wang Chu and Dr Xiao Weidi from Peking University contributed mass spectrometry data support. This work was funded by the National Natural Science Foundation of China, the National Key Basic Research Development Programme, the Shanghai Municipal Science and Technology Commission, the Shanghai Academy of Natural Sciences (SANS), and the Shanghai Branch of the Chinese Academy of Sciences.
Original link: https://www.nature.com/articles/s41589-025-01921-4






 Address:No.1308 Keyuan Road, Pudong District, Shanghai
Address:No.1308 Keyuan Road, Pudong District, Shanghai Phone:86-21-54740000
Phone:86-21-54740000 E-mail:zias@sjtu.edu.cn
E-mail:zias@sjtu.edu.cn