搜索


 Time:Aug 15, 2025
Time:Aug 15, 2025
On 15 August, Associate Professor Li Jian's team from Shanghai Jiao Tong University's Centre for Transformative Molecular Frontier Science, the State Key Laboratory of Chemical Biology-Based Material Innovation, and the Zhangjiang Advanced Research Institute, in collaboration with Professor Zhang Weidong and Associate Professor Wang Jinxin's team from the Naval Medical University and the Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, published a groundbreaking study in Science entitled: ‘Artificial farnesol epoxidase enables a concise synthesis of meroterpenoids’. Through directed evolution of P450BM3, the study yielded the artificial enzyme EPO6, capable of efficiently and selectively catalysing the epoxidation of the central alkene ring in farnesol. This enzyme maintained excellent enantiomeric (94% ee) and regioselectivity (93% r.s.) at gram-scale. By integrating this enzyme-catalysed technology with divergent synthetic approaches, the team reduced synthesis steps for 11 key natural products by 50–74%. This establishes a streamlined divergent synthetic pathway for efficient preparation of immunosuppressants and antitumour-active natural compounds.
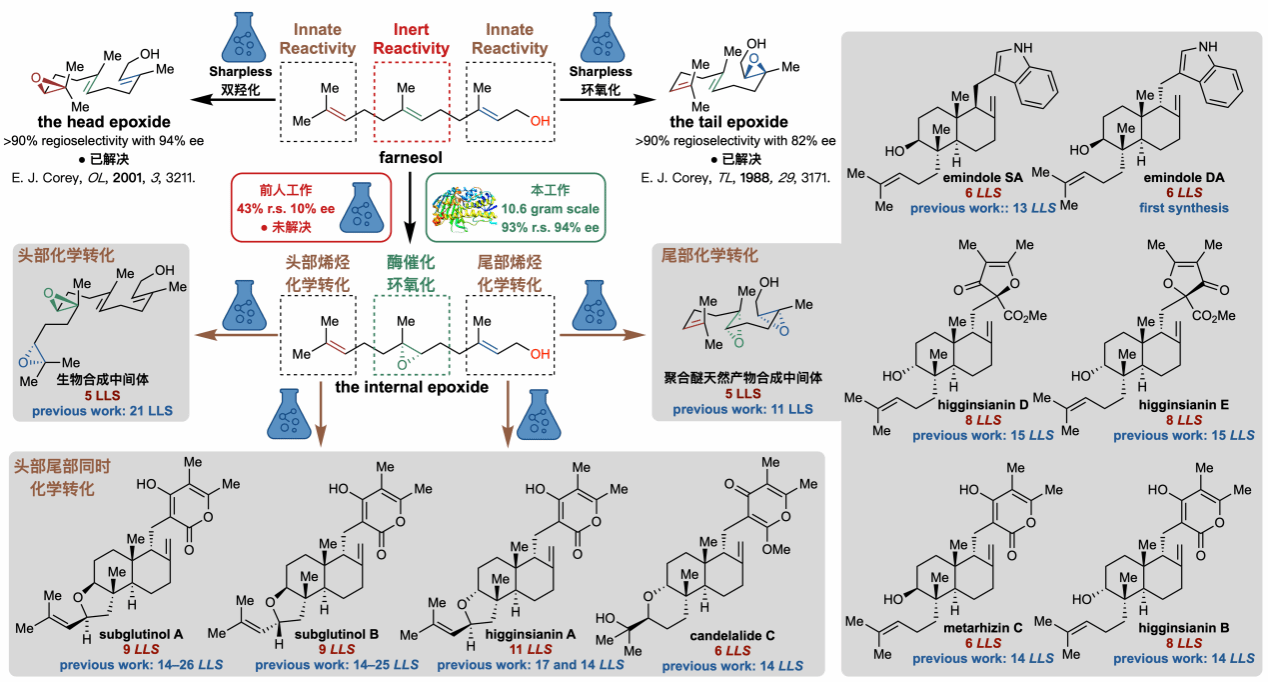
Farnesol serves as a crucial precursor for synthesising natural terpenoid products. However, the C6–C7 double bond at its molecular centre has long posed challenges for achieving high-precision selective epoxidation due to its substantial steric hindrance and lack of activating groups. The team screened their in-house enzyme library to identify the initial catalyst EPO1. Through 15 rounds of directed evolution involving saturation mutations of EPO1, they iteratively developed EPO6—a highly precise ‘molecular scissors’. EPO6 achieves 93% regioselectivity and 94% enantioselectivity for the central farnesol double bond, significantly outperforming existing chemical catalysis (43% regioselectivity, 10% enantioselectivity). Employing this biocatalyst, the team successfully synthesised 10 grams of chiral epoxide intermediates. This breakthrough achieved highly selective functionalisation of farnesol's most inert intramolecular double bond, enabling researchers to leverage the higher reactivity of its remaining double bonds for efficient, streamlined construction of diverse synthetic intermediates. Building upon this, the team validated a trimethylsilyl (TMS)-promoted mid-to-tail polyene tandem cyclisation strategy, effectively overcoming the cyclisation energy barrier posed by bulky side chains. This approach enabled the synthesis of 11 heteroterpenoid natural products, reducing the maximum linear steps (LLS) by up to 74% compared to conventional synthetic routes. Notably, Emindole DA was achieved through its first total synthesis, while Emindole SA was synthesised in just six steps. Subglutinol B was fully synthesised in just nine steps, significantly fewer than the 14–25 steps previously reported in the literature.
This work advances the paradigm shift in natural product and drug synthesis through chemical-biological synergy—transitioning from reliance on natural or existing enzyme catalysts and their simple modification towards a new phase where synthetic chemists precisely design and optimise ideal biocatalysts according to synthetic strategy requirements.
Associate Professor Li Jian, a resident scientist at the Centre for Frontier Science in Transformative Molecules and the Zhangjiang Advanced Research Institute, and Professor Zhang Weidong from the Naval Medical University and the Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, are the co-corresponding authors of this paper. Wang Jinxin, a visiting scholar at Shanghai Jiao Tong University and Associate Professor at the Naval Medical University, and Yin Yunpeng, a PhD candidate at the Centre for Frontier Science in Transformative Molecules, are the first authors. Zhang Quan, a doctoral candidate at the Centre for Frontier Science in Revolutionary Molecules, also made significant contributions to this research. This work was supported by the Shanghai Municipal Key Science and Technology Special Project, two National Key R&D Programmes (Approval Nos.: 2022YFC3502000; 2023YFA1506700), and two National Natural Science Foundation of China General Projects (Approval Nos.: 82430119; 82141203), the Central Universities Basic Research Fund (Approval Nos: 23X010301599; 24X010301678), and the Innovation Project of the Chinese Academy of Medical Sciences.
Paper link: https://www.science.org/doi/10.1126/science.adt2096
Author:
Li Jian's Team
Contributing Institution:
Frontiers Centre for Transformative Molecular Science






 Address:No.1308 Keyuan Road, Pudong District, Shanghai
Address:No.1308 Keyuan Road, Pudong District, Shanghai Phone:86-21-54740000
Phone:86-21-54740000 E-mail:zias@sjtu.edu.cn
E-mail:zias@sjtu.edu.cn