搜索


 Time:Oct 05, 2025
Time:Oct 05, 2025
On 2 October 2025 Beijing Time, the research group led by Professor Wen Liu from the School of Life Sciences and Technology at Shanghai Jiao Tong University, the Zhangjiang Advanced Research Institute, and the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, in collaboration with Research Fellows Lifeng Pan and Xiaosong Xue from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, and Professor Pengchen Ma from Xi'an Jiaotong University, achieved a significant breakthrough in the field of natural product biosynthesis. The findings, titled ‘Pseudokinases can catalyse peptide cyclisation through thioether crosslinking’, were published online in Nature Chemistry, ushering in new insights into pseudokinase research.
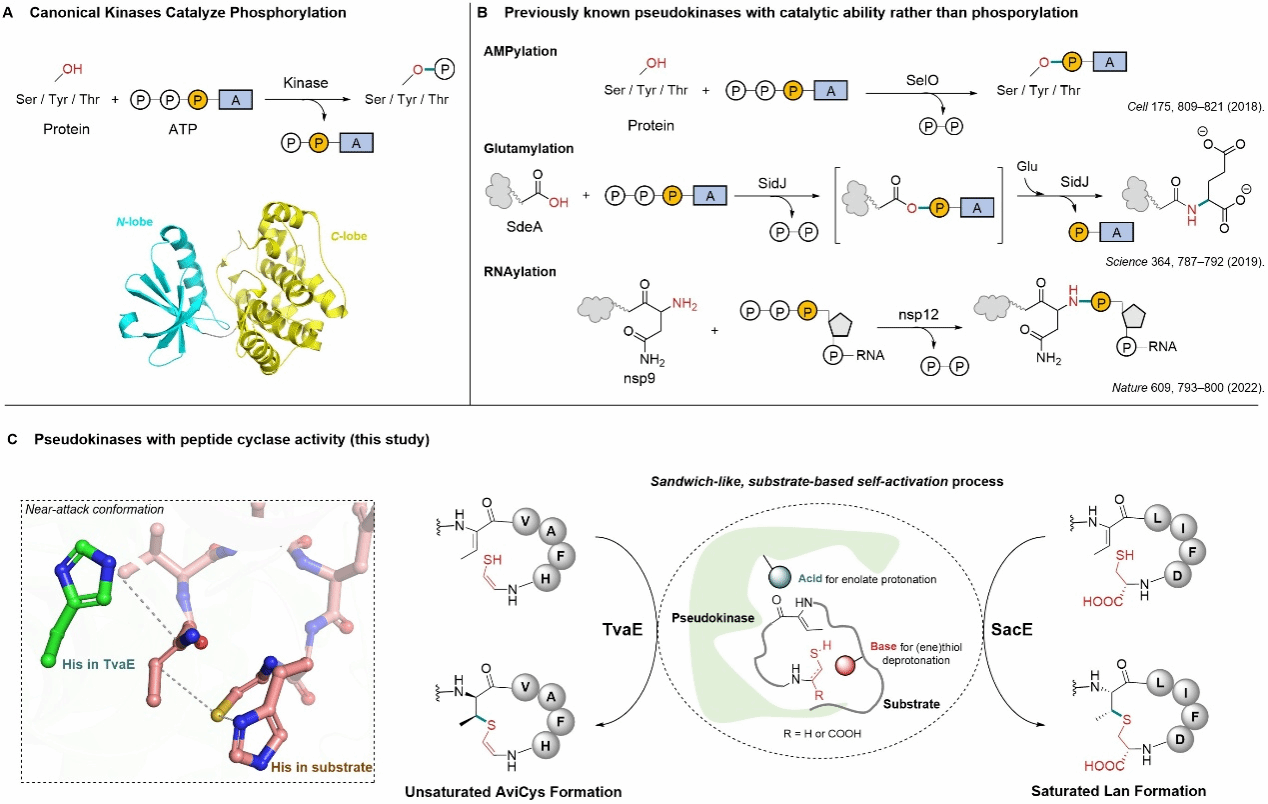
This study first demonstrated that bacterial pseudokinases can directly catalyse peptide cyclisation, forming unsaturated or saturated thioether crosslink structures. This breakthrough challenges the conventional understanding of pseudokinases as non-kinase catalysts, demonstrating the potential for functional reorganisation within the protein kinase superfamily during evolution. Through crystallography and multi-scale theoretical calculations, the team further proposed a unique ‘sandwich-like substrate-assisted’ mechanism, offering novel strategies for the efficient synthesis of cyclic peptide natural products.
The protein kinase superfamily, comprising over 70 families and millions of members, ranks among the most significant enzyme classes, with its hallmark function being ATP-dependent protein phosphorylation. Pseudokinases, lacking key residues, have long been regarded as lacking catalytic activity, serving solely in signal regulation and other roles. Previously, only a handful of pseudo-kinases were discovered to possess atypical functions, such as AMPylation, glutamylation, or RNA modification. However, these reactions all rely on the hydrolysis of high-energy phosphate bonds, and their functional scope remains highly limited.
In this study, the team focused on two classes of sulfur-rich bacterial cyclic peptides: thioamitides and lanthipepetides (lanthipeptides). Results revealed that in the thioamitide pathway, the pseudo-kinase TvaE catalyses Michael addition between terminal allyl mercaptan and dehydrated threonine residues, forming the AviCys cyclised structure; In the lanthipeptide pathway, pseudo-kinase SacE selectively catalyses the formation of saturated sulphide bonds, indicating that pseudo-kinase-mediated cyclisation functions are not isolated instances but may be prevalent across diverse natural products.
Crystallographic studies reveal that upon binding to its peptide substrate, TvaE completes the kinase structure, forming an ATP-independent active site. Combining molecular modelling, molecular dynamics simulations, and QM/MM calculations, the research team proposed a ‘sandwich-like substrate-assisted’ enzymatic reaction mechanism: The His residue on the peptide substrate acts as a base to deprotonate the thiol group, facilitating its addition to the upstream dehydrogenated amino acid. Subsequently, the His residue in the TvaE active site functions as an acid, completing the reaction through protonation to ultimately form a stereospecifically defined thioether cyclisation structure.
This discovery not only deepens our understanding of pseudokinase function but also provides novel tools and theoretical foundations for the biosynthesis of sulphur-rich cyclic peptides and the development of novel anti-cancer and anti-bacterial drugs.
The paper's co-first authors are Hu Ling, Li Miao, Sang Yueqian and Zhao Chengxin. The corresponding authors are Professor Liu Wen from the School of Life Sciences and Technology at Shanghai Jiao Tong University/Zhangjiang Institute for Advanced Studies/Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences; Research Fellows Pan Lifeng and Xue Xiaosong from the Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences; and Professor Ma Pengchen from Xi'an Jiaotong University. Shanghai Jiao Tong University is the primary institution for this work. This research received funding from the National Natural Science Foundation of China, the Ministry of Science and Technology, and the Shanghai Municipal Science and Technology Commission.
Expert Commentary (Professor Lei Xiaoguang, Peking University)
Due to their unique spatial structures and biological activities, cyclic peptides have demonstrated immense application potential in recent years across fields such as oral medications, peptide-drug conjugates, and diagnostic reagents. However, the efficient synthesis of cyclic peptide molecules remains highly challenging. Numerous cyclic peptide natural products exist in nature, with nonribosomal peptides (RiPPs) and post-translational modification peptides produced by ribosomes being the primary natural sources. The key cyclisation process in RiPP construction has long been a focal point and difficulty in related biosynthetic research. The discovery of cyclisation enzymes with unique catalytic mechanisms could facilitate the efficient construction of structurally diverse cyclic peptide molecules.
Recently, a collaborative study by Liu Wen's research team, published in Nature Chemistry, elucidated the fundamental scientific question of how the crucial intramolecular cyclisation processes occur in two important classes of natural products: thioamitides and lanthipeptides. This pioneering work uncovered an entirely novel catalytic mechanism for pseudo-kinases: a ‘sandwich-like substrate-assisted’ Michael addition reaction. This approach fundamentally diverges from previously known functional models of pseudo-kinases. The research elucidates the unique working mechanisms of the pseudokinases TvaE and SacE. Taking TvaE as an example, it binds to a leader peptide sequence to form a complete protein kinase folding structure, with this structural complementarity being crucial for TvaE's catalytic activity. Furthermore, the histidine residue (H86) within the substrate peptide directly participates in the catalytic reaction, acting as a base catalyst to activate the thiol group. This substrate-assisted catalysis mechanism is exceptionally rare within the kinase superfamily. This discovery challenges the long-held perception of pseudo-kinases as ‘non-catalytic,’ demonstrating the evolutionary ‘repurposing’ potential of protein kinase-like families. It also offers novel insights for the biosynthesis and engineering of cyclic peptide natural products.
Article link: https://doi.org/10.1038/s41557-025-01954-1






 Address:No.1308 Keyuan Road, Pudong District, Shanghai
Address:No.1308 Keyuan Road, Pudong District, Shanghai Phone:86-21-54740000
Phone:86-21-54740000 E-mail:zias@sjtu.edu.cn
E-mail:zias@sjtu.edu.cn